
Author:
Wendy Lovinger
The heart is a vital organ that pumps blood throughout the body, carrying oxygen and nutrients critical to organ function and sustaining life. It is, nevertheless, susceptible to disease. Heart disease touches the lives of almost everyone. The line between a healthy heart and an unhealthy heart is a fine one. Modern medicine has made significant advances in the technology needed to successfully intervene in the event of illness, but the technology can always be improved. One of the areas where improvements can continue to be made is mechanical heart valves.
Determining whether an implanted mechanical heart valve will open and close properly based on the actual blood flow usually requires patient participation, a high-risk proposition. Using computational fluid dynamics (CFD) to model mechanical heart valves, on the other hand, is a low-cost, low-risk method to evaluate device performance before performing an invasive procedure.
In this blog post, we explain how we simulated an idealized mechanical 3D heart valve with a small leaflet-to-blood density ratio using CONVERGE. We validated our results with the data from Banks et al., 2018.1
We modeled the motion of the mechanical heart valve with CONVERGE’s implicit fluid-structure interaction (FSI) solver. Because the density of blood is so close to the density of the heart valve, the added mass effect is significant, which can cause explicit FSI solvers to become unstable. CONVERGE’s implicit FSI solver can account for the additional inertial forces from the added mass effect. The implicit method tightly couples the CFD solver with the six degree-of-freedom rigid FSI solver, iterating between the two in a single-time step until the solution converges.
This implicit coupling allows us to predict the movement of an FSI object submerged in a fluid of a similar or higher density, such as a mechanical heart valve in blood. Figure 1 shows that our implicit FSI solver can accurately model how an idealized heart valve opens and closes for a range of leaflet-to-blood density ratios.
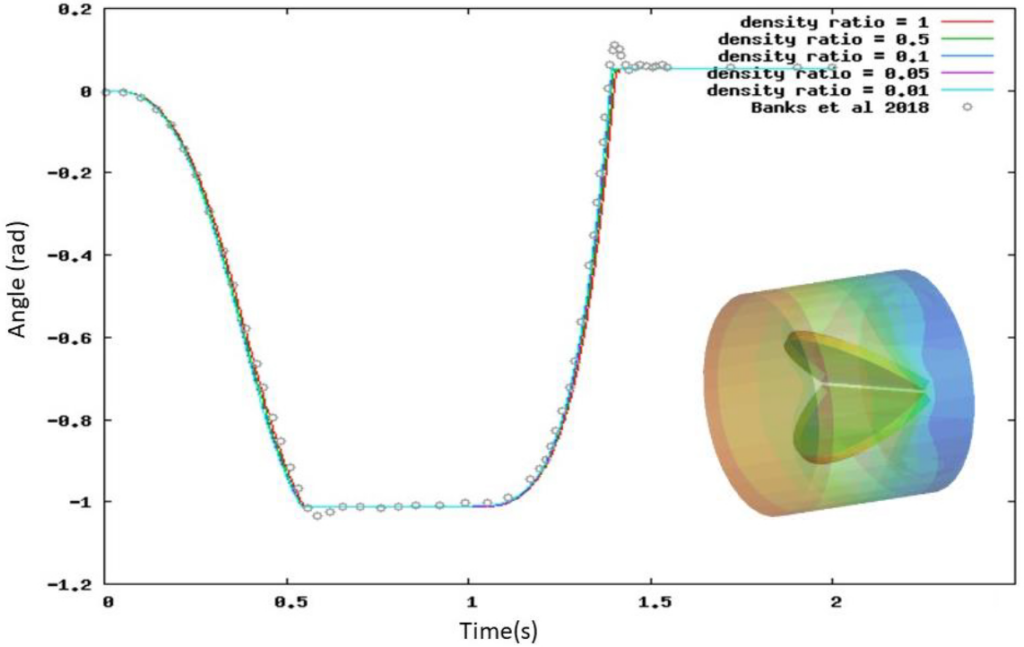
To capture the moving geometry of the mechanical heart valve, we used CONVERGE’s Cartesian cut-cell method with autonomous mesh generation. In some CFD solvers, creating an appropriate mesh for an FSI simulation can be challenging because you don’t know the motion profile ahead of time. In CONVERGE, the mesh is automatically regenerated near the FSI object at each time-step, easily accommodating the motion without any additional setup. We also deployed our Adaptive Mesh Refinement (AMR) to refine the grid in areas of high velocity gradient, which allows us to accurately capture the changes in velocity around the valve leaflet.
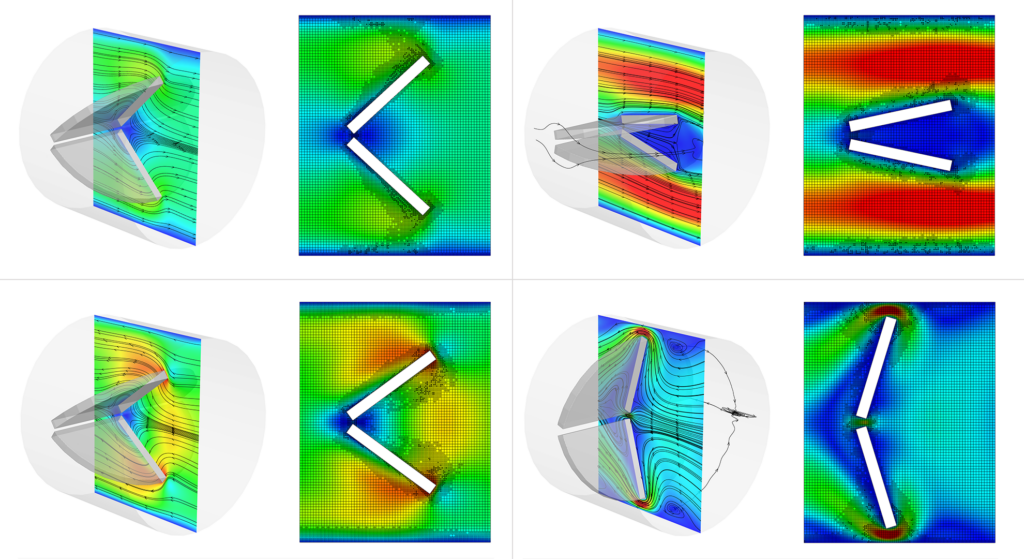
Figure 2 shows four velocity contour images at different stages of the heart valve opening and closing. CONVERGE’s AMR refines the grid only where the velocity changes the most and leaves the grid coarser where the flow is stagnant, greatly reducing computational expense.
Our results show you can accurately simulate an artificial heart valve with CONVERGE’s implicit FSI solver and autonomous meshing feature. Because CONVERGE allows you to easily modify your geometry, it is an excellent tool for evaluating the performance of different heart valve designs. Interested in finding out what other biomedical applications CONVERGE can be used for? Check out our biomedical webpage here!
[1] Banks, J.W., Henshaw, W.D., Schwendeman, D.W., and Tiang, Q., “A Stable Partitioned FSI Algorithm for Rigid Bodies and Incompressible Flow in Three Dimensions,” Journal of Computational Physics, 373, 455-492, 2018. DOI: 10.1016/j.jcp.2018.06.072


